
Sometimes there may be multiple compounds with that element on one side, so you'll need to use your best judgement and be prepared to go back and try the other options. 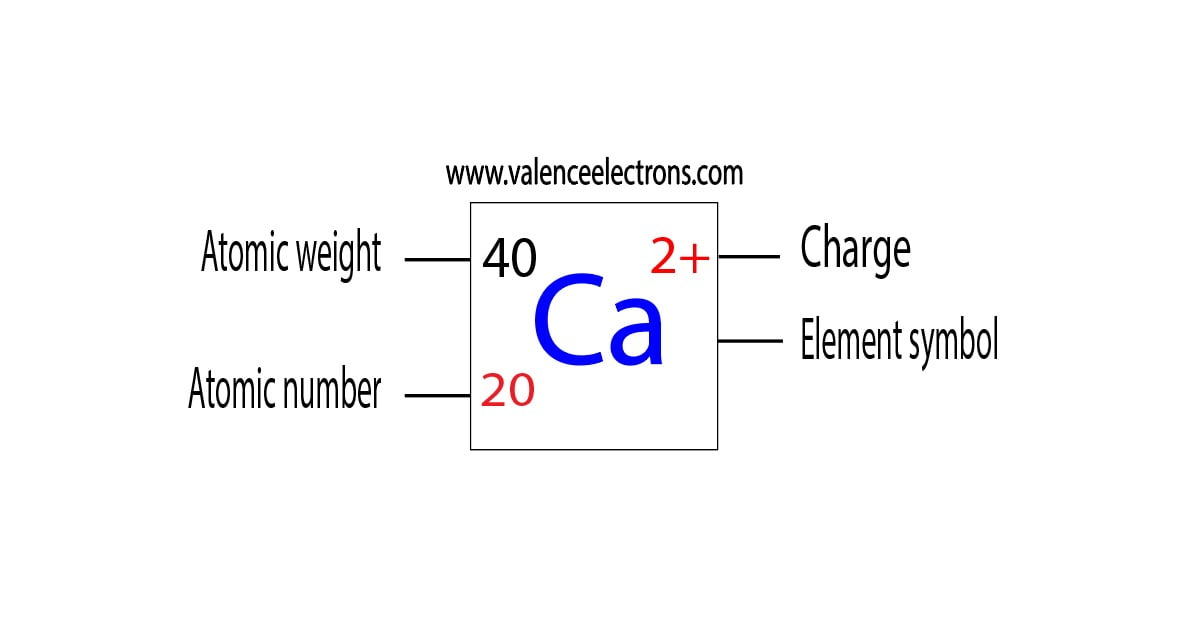
answered 03/11/19 Tutor 3.6 (18) Patient and Knowledgeable tutor specializing in organic chemistry. In print, we use roman numerals in parentheses to represent the charge on the ion so these. What is the charge of the calcium ion in CaCr2O7 Follow 1 Add comment Report 2 Answers By Expert Tutors Best Newest Oldest Nneamaka E. Thus, Fe 2 + is the iron two ion, while Fe 3 + is the iron three ion. If the element has more than one possible charge, the value of the charge comes after the element name and before the word ion. Transcribed image text: Refer to the following statement to answer questions 16-17: Calcium dichromate has the formula CaCr2O7 16. One can use the periodic table to predict whether an. (b) A sodium cation (Na+) has lost an electron, so it has one more proton (11) than electrons (10), giving it an overall positive charge, signified by a superscripted plus sign. Step 2: Multiply coefficients for compounds to balance out each elementįor each element that is not equal, try to balance it by adding more of it to the side with less. So Na + is the sodium ion Ca 2 + is the calcium ion. 1: (a) A sodium atom (Na) has equal numbers of protons and electrons (11) and is uncharged. 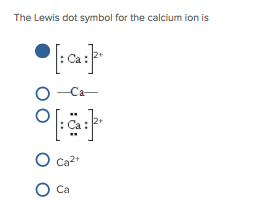
So, two chromium atoms have a total oxidation number of +12. Let x be the oxidation number of two chromium atoms. Molecular Weight 256.07 g/mol Computed by PubChem 2.1 (PubChem release 2021.05. There are seven oxygen atoms, so the total charge is 2714. Molecular Formula CaCr2O7 Synonyms Calcium dichromate Calcium bichromate 1 1 Calcium dichromate (VI) View More. The calcium atom has a radius of 197 pm and a Van der Waals. Oxygen is more electronegative than chromium, so itll take its usual 2 charge here. The number of electrons in each of Calciums shells is 2, 8, 8, 2 and its electron configuration is Ar4s 2. Step 1: Count the number of each element on the left and right hand sides Calcium ions are important messenger molecules in cells, which bind calcium-binding proteins to trigger many biochemical processes. Calcium (atomic symbol: Ca, atomic number: 20) is a Block S, Group 2, Period 4 element with an atomic weight of 40.078. When using the inspection method (also known as the trial-and-error method), this principle is used to balance one element at a time until both sides are equal and the chemical equation is balanced. To be balanced, every element in CaCl2 + K2Cr2O7 = KCl + CaCr2O7 must have the same number of atoms on each side of the equation. Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced. It turned out that what Caro and Frank had found was that that when calcium carbide is reacted with nitrogen at 1000 ◦C it forms calcium cyanamide (CaCN 2), (4.2.6).The law of conservation of mass states that matter cannot be created or destroyed, which means there must be the same number atoms at the end of a chemical reaction as at the beginning. Step 4: Substitute Coefficients and Verify Result. In 1898, one of their colleagues demonstrated that what was actually produced during the reaction at temperatures exceeding 1000 ☌ was not cyanide, as they had hoped. \).24), at the German chemical giant Badische Anilin- und Soda-Fabrik (BASF), were trying to make hydrogen cyanide (HCN) to use in its color dye business.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
